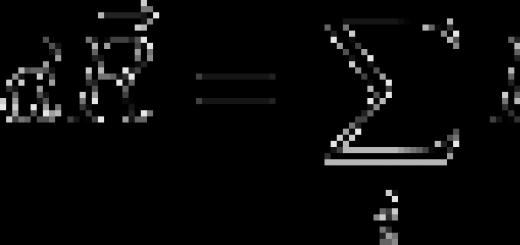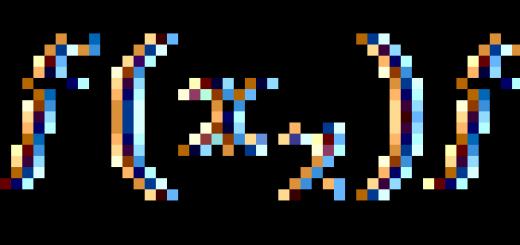Such an item school curriculum as chemistry causes numerous difficulties for most modern schoolchildren, few can determine the degree of oxidation in compounds. The greatest difficulties are for schoolchildren who study, that is, students of the main school (grades 8-9). Misunderstanding of the subject leads to the emergence of hostility among students to this subject.
Teachers identify a number of reasons for such a “dislike” of middle and high school students for chemistry: unwillingness to understand complex chemical terms, inability to use algorithms to consider a specific process, problems with mathematical knowledge. The Ministry of Education of the Russian Federation has made serious changes to the content of the subject. In addition, the number of hours for teaching chemistry was "cut down". This had a negative impact on the quality of knowledge in the subject, a decrease in interest in the study of the discipline.
What topics of the chemistry course are the most difficult for schoolchildren?
By new program to the course academic discipline"Chemistry" of the basic school includes several serious topics: the periodic table of elements of D. I. Mendeleev, classes inorganic substances, ion exchange. The hardest thing is for eighth graders to determine the degree of oxidation of oxides.
Placement rules
First of all, students should know that oxides are complex two-element compounds that include oxygen. A prerequisite for a binary compound to belong to the class of oxides is the second position of oxygen in this compound.
Algorithm for Acid Oxides
To begin with, we note that the degrees are numerical expressions of the valency of elements. Acid oxides formed by non-metals or metals with a valence of four to seven, the second in such oxides is necessarily oxygen.
In oxides, the valency of oxygen always corresponds to two; it can be determined by periodic table elements of D. I. Mendeleev. Such a typical non-metal as oxygen, being in the 6th group of the main subgroup of the periodic table, accepts two electrons in order to completely complete its external energy level. Non-metals in compounds with oxygen most often exhibit a higher valence, which corresponds to the number of the group itself. It is important to remember that the oxidation state chemical elements it is an indicator suggesting a positive (negative) number.
The non-metal at the beginning of the formula has a positive oxidation state. Non-metal oxygen is stable in oxides, its index is -2. In order to check the reliability of the arrangement of values in acid oxides, you will have to multiply all the numbers you set by the indices of a particular element. Calculations are considered reliable if the total sum of all the pluses and minuses of the set degrees is 0.

Compilation of two-element formulas
The oxidation state of the atoms of the elements gives a chance to create and record compounds from two elements. When creating a formula, for starters, both symbols are written side by side, be sure to put oxygen second. Above each of the recorded signs, the values \u200b\u200bof the oxidation states are prescribed, then between the numbers found is the number that will be divisible by both digits without any remainder. This indicator must be divided separately by the numerical value of the degree of oxidation, obtaining indices for the first and second components of the two-element substance. The highest oxidation state is numerically equal to the value of the highest valency of a typical non-metal, identical to the group number where the non-metal stands in PS.

Algorithm for setting numerical values in basic oxides
Oxides of typical metals are considered to be such compounds. They in all compounds have an oxidation state index of no more than +1 or +2. In order to understand what the oxidation state of a metal will be, you can use the periodic table. For metals of the main subgroups of the first group, this parameter is always constant, it is similar to the group number, that is, +1.
Metals of the main subgroup of the second group are also characterized by a stable oxidation state, numerically +2. The oxidation states of oxides, taking into account their indices (numbers), should add up to zero, since the chemical molecule is considered to be a neutral, charge-free particle.
Arrangement of oxidation states in oxygen-containing acids
Acids are complex substances, consisting of one or more hydrogen atoms, which are associated with some kind of acid residue. Given that oxidation states are numbers, some math skills are required to calculate them. Such an indicator for hydrogen (proton) in acids is always stable, it is +1. Next, you can specify the oxidation state for the negative oxygen ion, it is also stable, -2.
Only after these actions, it is possible to calculate the degree of oxidation of the central component of the formula. As a specific sample, consider the determination of the oxidation state of elements in sulfuric acid H2SO4. Considering that in the molecule of this complex substance contains two hydrogen protons, 4 oxygen atoms, we get an expression of this form +2+X-8=0. In order for the sum to form zero, sulfur will have an oxidation state of +6
Arrangement of oxidation states in salts
Salts are complex compounds consisting of metal ions and one or more acid residues. The procedure for determining the oxidation states of each of the constituents in a complex salt is the same as in oxygen-containing acids. Given that the oxidation state of the elements is digital indicator, it is important to correctly indicate the degree of oxidation of the metal.
If the salt-forming metal is located in the main subgroup, its oxidation state will be stable, corresponds to the group number, is a positive value. If the salt contains a metal of a similar subgroup of PS, it is possible to show different metals by the acid residue. After the oxidation state of the metal is set, put (-2), then the oxidation state of the central element is calculated using the chemical equation.
As an example, consider the determination of the oxidation states of elements in ( medium salt). NaNO3. The salt is formed by a metal of the main subgroup of group 1, therefore, the oxidation state of sodium will be +1. Oxygen in nitrates has an oxidation state of -2. To determine the numerical value of the degree of oxidation is the equation +1+X-6=0. Deciding given equation, we get that X should be +5, this is

Basic terms in OVR
For the oxidative as well as the reduction process, there are special terms that students are required to learn.
The oxidation state of an atom is its direct ability to attach to itself (donate to others) electrons from some ions or atoms.
Neutral atoms or charged ions are considered an oxidizing agent. chemical reaction accepting electrons.
The reducing agent will be uncharged atoms or charged ions, which in the process chemical interaction lose their own electrons.
Oxidation is presented as a procedure for donating electrons.
Reduction is associated with the acceptance of additional electrons by an uncharged atom or ion.
The redox process is characterized by a reaction during which the oxidation state of an atom necessarily changes. This definition allows you to understand how you can determine whether the reaction is OVR.

OVR Parsing Rules
Using this algorithm, you can arrange the coefficients in any chemical reaction.

Target: Continue studying valency. Give the concept of oxidation state. Consider the types of oxidation states: positive, negative, zero value. Learn to correctly determine the oxidation state of an atom in a compound. To teach methods of comparison and generalization of the concepts being studied; develop skills and abilities in determining the degree of oxidation by chemical formulas; continue developing skills independent work; promote the development of logical thinking. To form a sense of tolerance (tolerance and respect for other people's opinions) of mutual assistance; to carry out aesthetic education (through the design of the board and notebooks, when using presentations).
During the classes
I. Organizing time
Checking students for class.
II. Preparing for the lesson.
By the lesson you will need: Periodic system of D.I. Mendeleev, textbook, workbooks, pens, pencils.
III. Checking homework.
Frontal survey, some will work at the board on cards, conducting a test, and summing up this stage will be an intellectual game.
1. Work with cards.
1 card
Define mass fractions(%) carbon and oxygen in carbon dioxide (CO 2 ) .
2 card
Determine the type of bond in the H 2 S molecule. Write the structural and electronic formulas of the molecule.
2. Frontal survey
- What is a chemical bond?
- What types of chemical bonds do you know?
- What bond is called a covalent bond?
- What covalent bonds are isolated?
- What is valence?
- How do we define valency?
- Which elements (metals and non-metals) have variable valency?
3. Testing
1. Which molecules have non-polar covalent bonds?
2 . Which molecule forms a triple bond when a covalent-nonpolar bond is formed?
3 . What are positively charged ions called?
A) cations
B) molecules
B) anions
D) crystals
4. In which order are the substances of an ionic compound located?
A) CH 4, NH 3, Mg
B) CI 2, MgO, NaCI
B) MgF 2, NaCI, CaCI 2
D) H 2 S, HCI, H 2 O
5 . Valency is determined by:
A) by group number
B) by the number of unpaired electrons
B) by type chemical bond
D) by period number.
4. Intellectual game"Tic Tac Toe »
Find substances with a covalent-polar bond.
IV. Learning new material
The oxidation state is an important characteristic of the state of an atom in a molecule. Valence is determined by the number of unpaired electrons in an atom, orbitals with unshared electron pairs, only in the process of excitation of the atom. The highest valency of an element is usually equal to the group number. The degree of oxidation in compounds with different chemical bonds is formed unequally.
How is the oxidation state formed in molecules with different chemical bonds?
1) In compounds with an ionic bond, the oxidation state of the elements is equal to the charges of the ions.
2) In compounds with a covalent non-polar bond (in molecules simple substances) the oxidation state of the elements is 0.
H 2 0 , CI 2 0 , F 2 0 , S 0 , AI 0
3) For molecules with a covalent-polar bond, the degree of oxidation is determined similarly to molecules with an ionic chemical bond.
The oxidation state of the element - this is the conditional charge of its atom, in a molecule, if we assume that the molecule consists of ions.
The oxidation state of an atom, in contrast to the valency, has a sign. It can be positive, negative or zero.
Valency is indicated by Roman numerals on top of the element symbol:
II |
I |
IV |
Fe |
Cu |
S, |
and the oxidation state is indicated by Arabic numerals with a charge above the element symbols ( Mg +2 , Ca +2 ,Na +1,CIˉ¹).
A positive oxidation state is equal to the number of electrons donated to these atoms. An atom can donate all valence electrons (for the main groups, these are electrons of the outer level) corresponding to the number of the group in which the element is located, while showing the highest oxidation state (with the exception of OF 2). For example: the highest oxidation state of the main subgroup of group II is +2 ( Zn +2) A positive degree is shown by both metals and non-metals, except for F, He, Ne. For example: C+4 ,Na+1 , Al+3
The negative oxidation state is equal to the number of electrons accepted by a given atom, it is shown only by non-metals. Atoms of non-metals attach as many electrons as they are not enough to complete the external level, while showing a negative degree.
For elements of the main subgroups of IV-VII groups, the minimum oxidation state is numerically equal to
For example:
The value of the oxidation state between the highest and lowest oxidation states is called intermediate:
Higher |
Intermediate |
Inferior |
C +3, C +2, C 0, C -2 |
||
In compounds with a covalent non-polar bond (in molecules of simple substances), the oxidation state of the elements is 0: H 2 0 , FROMI 2 0 , F 2 0 , S 0 , AI 0
To determine the oxidation state of an atom in a compound, a number of provisions should be taken into account:
1. Oxidation stateFin all compounds is equal to "-1".Na +1 F -1 , H +1 F -1
2. The oxidation state of oxygen in most compounds is (-2) exception: OF 2 , where the oxidation state is O +2F -1
3. Hydrogen in most compounds has an oxidation state of +1, except for compounds with active metals, where the oxidation state is (-1): Na +1 H -1
4. The degree of oxidation of metals of the main subgroupsI, II, IIIgroups in all compounds is +1,+2,+3.
Elements with a constant oxidation state are:
BUT) alkali metals(Li, Na, K, Pb, Si, Fr) - oxidation state +1
B) elements of the II main subgroup of the group except (Hg): Be, Mg, Ca, Sr, Ra, Zn, Cd - oxidation state +2
C) element of group III: Al - oxidation state +3
Algorithm for compiling a formula in compounds:
1 way
1 . The element with the lowest electronegativity is listed first, the element with the highest electronegativity is listed second.
2 . The element written in the first place has a positive charge "+", and in the second with a negative charge "-".
3 . Specify the oxidation state for each element.
4 . Find the total multiple of the oxidation states.
5. Divide the least common multiple by the value of the oxidation states and assign the resulting indices to the bottom right after the symbol of the corresponding element.

6. If the oxidation state is even - odd, then they become next to the symbol at the bottom right of the cross - crosswise without the sign "+" and "-":
![]()
7. If the oxidation state has an even value, then they must first be reduced by smallest value oxidation states and put a cross - crosswise without the sign "+" and "-": C +4 O -2

2 way
1 . Let's denote the oxidation state of N through X, indicate the oxidation state of O: N 2 xO 3 -2
2 . Determine the sum of negative charges, for this, the oxidation state of oxygen is multiplied by the oxygen index: 3 (-2) \u003d -6
3 .For the molecule to be electrically neutral, you need to determine the sum of positive charges: X2 \u003d 2X
4 .Make an algebraic equation:
N 2 + 3 O 3 –2
V. Anchoring
1) Carrying out the fixing of the topic by the game, which is called "Snake".
Rules of the game: the teacher distributes cards. Each card has one question and one answer to another question.
The teacher starts the game. He reads out the question, the student who has the answer to my question raises his hand and says the answer. If the answer is correct, then he reads his question and the student who has the answer to this question raises his hand and answers, etc. A snake of correct answers is formed.
- How and where is the oxidation state of an atom of a chemical element indicated?
Answer: an Arabic numeral above the element symbol with charge "+" and "-". - What types of oxidation states are distinguished from atoms of chemical elements?
Answer: intermediate - What degree does metals exhibit?
Answer: positive, negative, zero. - What degree show simple substances or molecules with a non-polar covalent bond.
Answer: positive - What charge do cations and anions have?
Answer: null. - What is the name of the oxidation state that stands between the positive and negative oxidation states.
Answer: positive, negative
2) Write formulas of substances consisting of the following elements
- N and H
- R&O
- Zn and Cl
3) Find and cross out substances that do not have a variable oxidation state.
Na, Cr, Fe, K, N, Hg, S, Al, C
VI. Summary of the lesson.
Rating with comments
VII. Homework
§23, p.67-72, task after §23-p. 72 No. 1-4 to complete.
DEFINITION
Oxidation state is a quantitative assessment of the state of an atom of a chemical element in a compound, based on its electronegativity.
She accepts both positive and negative values. To indicate the oxidation state of an element in a compound, you need to put an Arabic numeral with the corresponding sign ("+" or "-") above its symbol.
It should be remembered that the degree of oxidation is a quantity that does not have physical sense, since it does not reflect the real charge of the atom. However, this concept is very widely used in chemistry.
Table of the oxidation state of chemical elements
The maximum positive and minimum negative oxidation states can be determined using the Periodic Table of D.I. Mendeleev. They are equal to the number of the group in which the element is located, and the difference between the value of the "highest" oxidation state and the number 8, respectively.
If we consider chemical compounds more specifically, in substances with non-polar bonds, the oxidation state of the elements is zero (N 2, H 2, Cl 2).
The oxidation state of metals in the elementary state is zero, since the distribution of electron density in them is uniform.
In simple ionic compounds, the oxidation state of their constituent elements is electric charge, since during the formation of these compounds there is an almost complete transition of electrons from one atom to another: Na +1 I -1, Mg +2 Cl -1 2, Al +3 F -1 3, Zr +4 Br -1 4.
When determining the oxidation state of elements in compounds with polar covalent bonds compare their electronegativity values. Since, during the formation of a chemical bond, electrons are displaced to atoms of more electronegative elements, the latter have a negative oxidation state in compounds.
There are elements for which only one value of the oxidation state is characteristic (fluorine, metals of IA and IIA groups, etc.). Fluorine, characterized highest value electronegativity, in compounds it always has a constant negative oxidation state (-1).
Alkaline and alkaline earth elements, which are characterized by a relatively low value of electronegativity, always have a positive oxidation state, equal to (+1) and (+2), respectively.
However, there are also such chemical elements, which are characterized by several values of the degree of oxidation (sulfur - (-2), 0, (+2), (+4), (+6), etc.).
In order to make it easier to remember how many and what oxidation states are characteristic of a particular chemical element, tables of the oxidation states of chemical elements are used, which look like this:
|
Serial number |
Russian / English title |
chemical symbol |
Oxidation state |
|
Hydrogen |
|||
|
Helium / Helium |
|||
|
Lithium / Lithium |
|||
|
Beryllium / Beryllium |
|||
|
(-1), 0, (+1), (+2), (+3) |
|||
|
Carbon / Carbon |
(-4), (-3), (-2), (-1), 0, (+2), (+4) |
||
|
Nitrogen / Nitrogen |
(-3), (-2), (-1), 0, (+1), (+2), (+3), (+4), (+5) |
||
|
Oxygen / Oxygen |
(-2), (-1), 0, (+1), (+2) |
||
|
Fluorine / Fluorine |
|||
|
Sodium |
|||
|
Magnesium / Magnesium |
|||
|
Aluminum |
|||
|
Silicon / Silicon |
(-4), 0, (+2), (+4) |
||
|
Phosphorus / Phosphorus |
(-3), 0, (+3), (+5) |
||
|
Sulfur |
(-2), 0, (+4), (+6) |
||
|
Chlorine / Chlorine |
(-1), 0, (+1), (+3), (+5), (+7), rarely (+2) and (+4) |
||
|
Argon / Argon |
|||
|
Potassium / Potassium |
|||
|
Calcium / Calcium |
|||
|
Scandium / Scandium |
|||
|
Titanium / Titanium |
(+2), (+3), (+4) |
||
|
Vanadium / Vanadium |
(+2), (+3), (+4), (+5) |
||
|
Chromium / Chromium |
(+2), (+3), (+6) |
||
|
Manganese / Manganese |
(+2), (+3), (+4), (+6), (+7) |
||
|
Iron / Iron |
(+2), (+3), rarely (+4) and (+6) |
||
|
Cobalt / Cobalt |
(+2), (+3), rarely (+4) |
||
|
Nickel / Nickel |
(+2), rarely (+1), (+3) and (+4) |
||
|
Copper |
+1, +2, rare (+3) |
||
|
Gallium / Gallium |
(+3), rare (+2) |
||
|
Germanium / Germanium |
(-4), (+2), (+4) |
||
|
Arsenic / Arsenic |
(-3), (+3), (+5), rarely (+2) |
||
|
Selenium / Selenium |
(-2), (+4), (+6), rarely (+2) |
||
|
Bromine / Bromine |
(-1), (+1), (+5), rarely (+3), (+4) |
||
|
Krypton / Krypton |
|||
|
Rubidium / Rubidium |
|||
|
Strontium / Strontium |
|||
|
Yttrium / Yttrium |
|||
|
Zirconium / Zirconium |
(+4), rarely (+2) and (+3) |
||
|
Niobium / Niobium |
(+3), (+5), rarely (+2) and (+4) |
||
|
Molybdenum / Molybdenum |
(+3), (+6), rarely (+2), (+3) and (+5) |
||
|
Technetium / Technetium |
|||
|
Ruthenium / Ruthenium |
(+3), (+4), (+8), rarely (+2), (+6) and (+7) |
||
|
Rhodium |
(+4), rarely (+2), (+3) and (+6) |
||
|
Palladium / Palladium |
(+2), (+4), rarely (+6) |
||
|
Silver / Silver |
(+1), rarely (+2) and (+3) |
||
|
Cadmium / Cadmium |
(+2), rare (+1) |
||
|
Indium / Indium |
(+3), rarely (+1) and (+2) |
||
|
Tin / Tin |
(+2), (+4) |
||
|
Antimony / Antimony |
(-3), (+3), (+5), rarely (+4) |
||
|
Tellurium / Tellurium |
(-2), (+4), (+6), rarely (+2) |
||
|
(-1), (+1), (+5), (+7), rarely (+3), (+4) |
|||
|
Xenon / Xenon |
|||
|
Cesium / Cesium |
|||
|
Barium / Barium |
|||
|
Lanthanum / Lanthanum |
|||
|
Cerium / Cerium |
(+3), (+4) |
||
|
Praseodymium / Praseodymium |
|||
|
Neodymium / Neodymium |
(+3), (+4) |
||
|
Promethium / Promethium |
|||
|
Samaria / Samarium |
(+3), rare (+2) |
||
|
Europium / Europium |
(+3), rare (+2) |
||
|
Gadolinium / Gadolinium |
|||
|
Terbium / Terbium |
(+3), (+4) |
||
|
Dysprosium / Dysprosium |
|||
|
Holmium / Holmium |
|||
|
Erbium / Erbium |
|||
|
Thulium / Thulium |
(+3), rare (+2) |
||
|
Ytterbium / Ytterbium |
(+3), rare (+2) |
||
|
Lutetium / Lutetium |
|||
|
Hafnium / Hafnium |
|||
|
Tantalum / Tantalum |
(+5), rarely (+3), (+4) |
||
|
Tungsten / Tungsten |
(+6), rare (+2), (+3), (+4) and (+5) |
||
|
Rhenium / Rhenium |
(+2), (+4), (+6), (+7), rarely (-1), (+1), (+3), (+5) |
||
|
Osmium / Osmium |
(+3), (+4), (+6), (+8), rarely (+2) |
||
|
Iridium / Iridium |
(+3), (+4), (+6), rarely (+1) and (+2) |
||
|
Platinum / Platinum |
(+2), (+4), (+6), rarely (+1) and (+3) |
||
|
Gold / Gold |
(+1), (+3), rarely (+2) |
||
|
Mercury / Mercury |
(+1), (+2) |
||
|
Waist / Thallium |
(+1), (+3), rarely (+2) |
||
|
Lead / Lead |
(+2), (+4) |
||
|
Bismuth / Bismuth |
(+3), rarely (+3), (+2), (+4) and (+5) |
||
|
Polonium / Polonium |
(+2), (+4), rarely (-2) and (+6) |
||
|
Astatine / Astatine |
|||
|
Radon / Radon |
|||
|
Francium / Francium |
|||
|
Radium / Radium |
|||
|
Actinium / Actinium |
|||
|
Thorium / Thorium |
|||
|
Proactinium / Protactinium |
|||
|
Uranus / Uranium |
(+3), (+4), (+6), rarely (+2) and (+5) |
Examples of problem solving
EXAMPLE 1
- The oxidation state of phosphorus in phosphine is (-3), and in phosphoric acid - (+5). Change in the oxidation state of phosphorus: +3 → +5, i.e. the first answer.
- The oxidation state of a chemical element in a simple substance is zero. The oxidation state of phosphorus in the oxide composition P 2 O 5 is equal to (+5). Change in the oxidation state of phosphorus: 0 → +5, i.e. third answer.
- The oxidation state of phosphorus in an acid of composition HPO 3 is (+5), and H 3 PO 2 is (+1). Change in the oxidation state of phosphorus: +5 → +1, i.e. fifth answer.
EXAMPLE 2
| The task | The oxidation state (-3) carbon has in the compound: a) CH 3 Cl; b) C 2 H 2 ; c) HCOH; d) C 2 H 6 . |
| Decision | In order to give a correct answer to the question posed, we will alternately determine the degree of carbon oxidation in each of the proposed compounds. a) the oxidation state of hydrogen is (+1), and chlorine - (-1). We take for "x" the degree of oxidation of carbon: x + 3×1 + (-1) =0; The answer is incorrect. b) the oxidation state of hydrogen is (+1). We take for "y" the degree of oxidation of carbon: 2×y + 2×1 = 0; The answer is incorrect. c) the oxidation state of hydrogen is (+1), and oxygen - (-2). Let's take for "z" the oxidation state of carbon: 1 + z + (-2) +1 = 0: The answer is incorrect. d) the oxidation state of hydrogen is (+1). Let's take for "a" the oxidation state of carbon: 2×a + 6×1 = 0; Correct answer. |
| Answer | Option (d) |
Table. Degrees of oxidation of chemical elements.
Table. Degrees of oxidation of chemical elements.
Oxidation state is the conditional charge of the atoms of a chemical element in a compound, calculated from the assumption that all bonds are of the ionic type. Oxidation states can be positive, negative, or zero, so algebraic sum the oxidation states of elements in a molecule, taking into account the number of their atoms, is 0, and in an ion - the charge of the ion.
|
Table: Elements with constant oxidation states. |
|
Table. The oxidation states of chemical elements in alphabetical order.
|
Table. The oxidation states of chemical elements by number.
|
Article rating:
The degree of oxidation is a conditional value used to record redox reactions. To determine the degree of oxidation, a table of oxidation of chemical elements is used.
Meaning
The oxidation state of basic chemical elements is based on their electronegativity. The value is equal to the number of electrons displaced in the compounds.
The oxidation state is considered positive if the electrons are displaced from the atom, i.e. the element donates electrons in the compound and is a reducing agent. These elements include metals, their oxidation state is always positive.
When an electron is displaced towards an atom, the value is considered negative, and the element is considered an oxidizing agent. The atom accepts electrons before the completion of the external energy level. Most non-metals are oxidizing agents.
Simple substances that do not react always have a zero oxidation state.

Rice. 1. Table of oxidation states.
In the compound, a non-metal atom with a lower electronegativity has a positive oxidation state.
Definition
You can determine the maximum and minimum oxidation state (how many electrons an atom can give and take) using the periodic table of Mendeleev.
Max Degree equal to the number of the group in which the element is located, or the number of valence electrons. The minimum value is determined by the formula:
No. (groups) - 8.

Rice. 2. Periodic table.
Carbon is in the fourth group, therefore, its highest oxidation state is +4, and the lowest is -4. The maximum oxidation state of sulfur is +6, the minimum is -2. Most non-metals always have a variable - positive and negative - oxidation state. The exception is fluorine. Its oxidation state is always -1.
It should be remembered that this rule does not apply to alkali and alkaline earth metals of groups I and II, respectively. These metals have a constant positive oxidation state - lithium Li +1, sodium Na +1, potassium K +1, beryllium Be +2, magnesium Mg +2, calcium Ca +2, strontium Sr +2, barium Ba +2. Other metals may show varying degrees oxidation. The exception is aluminum. Despite being in group III, its oxidation state is always +3.

Rice. 3. Alkali and alkaline earth metals.
Of group VIII, only ruthenium and osmium can exhibit the highest oxidation state +8. Gold and copper, which are in group I, exhibit oxidation states of +3 and +2, respectively.
Recording
To correctly record the oxidation state, you should remember a few rules:
- inert gases do not react, so their oxidation state is always zero;
- in connections variable degree oxidation depends on variable valency and interaction with other elements;
- hydrogen in compounds with metals exhibits a negative oxidation state - Ca +2 H 2 −1, Na +1 H −1;
- oxygen always has an oxidation state of -2, except for oxygen fluoride and peroxide - O +2 F 2 -1, H 2 +1 O 2 -1.
What have we learned?
The oxidation state is a conditional value showing how many electrons an atom of an element has received or given away in a compound. The value depends on the number of valence electrons. Metals in compounds always have a positive oxidation state, i.e. are restorers. For alkali and alkaline earth metals, the oxidation state is always the same. Non-metals, except for fluorine, can take positive and negative oxidation states.